There is likely no other area of medical research with more potential for reducing the suffering of chronic disease than the billions of tiny passengers inside our guts.
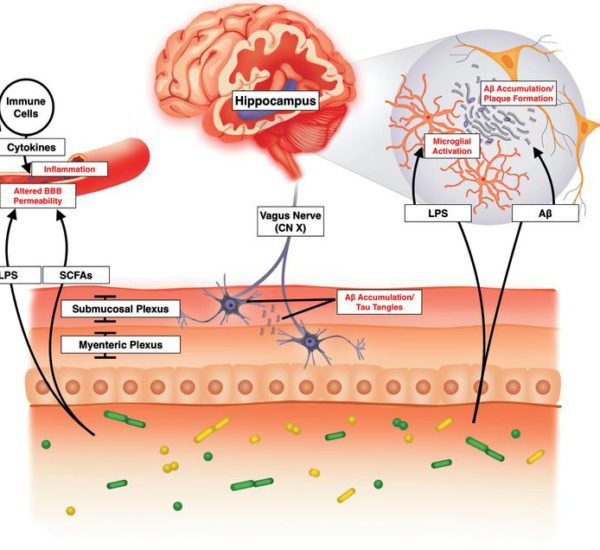
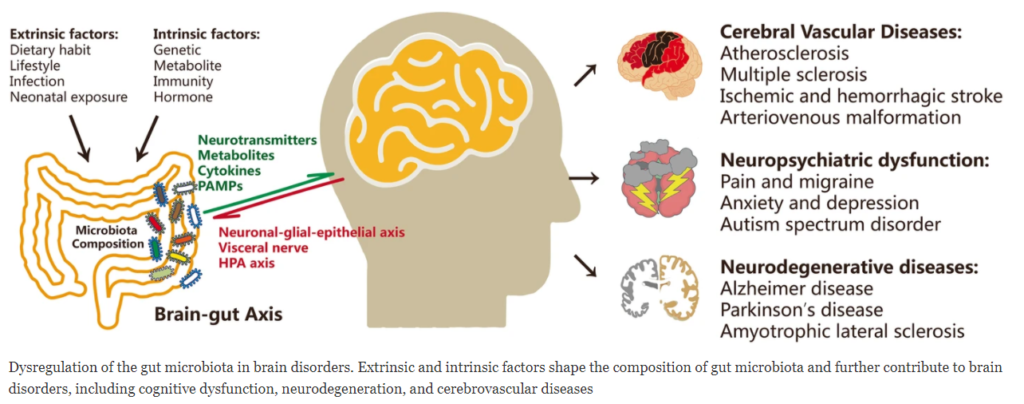
There is increasing evidence showing that the dynamic changes in the gut microbiota can alter brain physiology and behavior. Cognition was originally thought to be regulated only by the central nervous system. However, it is now clear that the gut-resident bacteria of the gastrointestinal tract, regulate and influence cognitive dysfunction as well as the process of neurodegeneration and cerebrovascular diseases.
Controllable factors such as diet can have a massive effect on the composition of the microbiota for good or ill. Microbes release molecules that trigger inflammation throughout the central nervous system.
Many recent studies have shown that neuroinflammation contributes greatly to the development of neurological disorders such as
- pain
- depression
- anxiety
- autism
- Alzheimer’s diseases
- Parkinson’s disease
- stroke
The most critical mechanisms of action in the development of downstream neurological dysfunction include:
- Change of blood-brain barrier permeability allowing harmful bacteria and proteins to enter and damage the brain
- brain vascular physiology
- brain structure
The following table illustrates the results of recent rodent studies and the effects of the presence or absence of strains of bacteria in the gut.
| Animal | Model type | Phenotype and effect (vs. controls) |
|---|---|---|
| NMRI mice | Germ-free | Increased motor activity and reduced anxiety [61] |
| Wistar rat | Application of probiotics: Lactobacillus helveticus R0052 and Bifidobacterium longum R0175 | Decreased stress-induced gastrointestinal discomfort and anxiolytic effect [118] |
| Balb/c mice | Application of probiotics: Lactobacillus rhamnosus (JB-1) | Increase of corticosterone, GABA receptor, N-acetyl aspartate and glutamate; reduced stress-induced corticosterone and anxiety- and depression-related behavior [119, 120] |
| Balb/c mice | Germ-free, SPF and B. infantis mice | GF mice exhibited exaggerated HPA stress response and decreased BDNF compared to SPF mice, but can be reversed with B. infantis [121] |
| Swiss Webster mice | Germ-free | GF mice have a anxiolytic behavior, increased BDNF and reduced serotonin 1A receptor in dentate gyrus of the hippocampus [122] |
| Balb/c mice and AKH mice | T. muris infection and application of probiotics B. longum NCC3001 | Infection with T. muris induced anxiety-like behavior and decreased level of BNDF. Treatment with B. longum reverses the effect and normalizes BDNF level [123] |
| Balb/c mice and NIH mice | Germ-free and SPF with antibiotics | Increased exploratory behavior and hippocampal expression of BDNF in hippocampus [124] |
| Stress-sensitive F344 Rat | Germ-free and SPF | Decreased social interaction in GF mice [125] |
| Swiss Webster mice | Germ-free | Significant social impairments and decreased social preference in GF mice [126] |
| Wistar rat | Application of antibiotic | Reduced social interactions in offsprings when antibiotic is applied peri-conceptionally [127] |
| Swiss Webster mice | Germ-free | Increased exploratory in GF mice [122] |
| Swiss Webster mice | Application of antibiotic | Increased exploratory behavior in antibiotic-treated mice [126] |
| C57BL/6 mice | Application of antibiotic | Decreased working memory in antibiotic-treated SPF mice; no effect on spatial memory [128] |
| Swiss Webster mice | Germ-free | Decreased working memory in GF mice [129] |
| C57BL/6 mice | SPF | Decreased working memory in antibiotic-treated SPF mice [130] |
| C57BL/6 mice and CF1 mice | Citrobacter rodentidum infection | Increase anxiety and memory dysfunction [129, 131] |
The goal of the FEDMI Microbiome Optimization Initiative is to extend this research by promoting the collection and analysis of:
- microbiome samples
- data on the severity and incidence of various chronic illness for those donors
Once this data is available it can be used to determine the optimal ratios of various strains of bacteria to alleviate specific diseases.
At this time, the focus will be directed at quantifying the effectiveness of various techniques of adjusting the ratio of various species including:
- prebiotic and probiotic supplementation
- dietary modifications
- fecal microbiota transplantation